 (source)
(source)
|
William C. Kelly
(21 Aug 1811 - 11 Feb 1888)
American ironmaster who invented the pneumatic process of making steel. While a Kentucky manufacturer of iron kettles, he discovered that cold air blown on red-hot iron caused the metal to become white-hot by igniting the carbon and thus eliminating impurities. He defended his belated patent on the process, and won, against the earlier-filed U.S. patent of Sir Henry Bessemer of Great Britain.
|
To all whom it may concern:
Be it known that I, WILLIAM KELLY, of Lyon county, Kentucky, have discovered a new and Improved Method of Treating Iron, by which I am enabled to refine and decarbonize crude pig metal or iron in a fluid state without the use of fuel.
The nature of my invention consists in the discovery that the carbon mechanically combined with iron, and which is burned from the fuel while in the process of smelting in the blast-furnace, is of itself (the carbon) sufficient, when combined with the oxygen of the air, to create heat enough and of sufficient intensity to keep melted pig iron or metal in a fluid or lively state long enough to carry the metal through, without chilling, all the various manipulations of refining without the aid of any other heat than that obtained by the above - described chemical union of oxygen and carbon.
I am aware that it is well known that oxygen and carbon, when combined or brought together, produce heat; but it is not known that the amount of these chemical properties in air and iron is the required quantity necessary to produce heat sufficient to carry out the practical refining of crude pig-iron; hence the prevailing opinion among iron-workers that a blast of cold air driven into a body of liquid iron would chill it. Therefore, when iron is worked in the finery or run-out fire the presence of heat from other sources is deemed indispensable to prevent the chilling of the iron. The finery or run-out fire is usually open on three sides, sometimes closed except at top to receive the charge of coal and iron.
A new furnace or cupola to work iron under my new process must be constructed as close as possible to prevent a loss of iron which would occur on account of its violent boiling,
during which particles are thrown up and adhere to the sides and top of the chamber, but which during the process are remelted and flow down to the mass in the bottom. In the finery or run-out this loss is prevented by the iron being covered by fuel. It is also first charged with metal in a solid state. In my process the metal is taken in a fluid state from the blast-furnace and put in the cupola or furnace. In the finery or run-out the iron is brought to a fluid state by mixing it with large quantities of fuel, and when melted falls to the bottom of the finery, where it is decarbonized by strong blasts of air in connection with the fuel. In my process no fuel of any kind is used or required, as I rely exclusively on the heat created or generated by the chemical union of oxygen in the air and carbon in the iron.
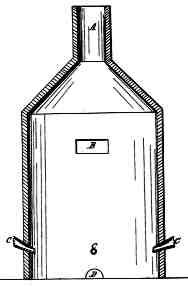
In the accompanying drawings, Figure 1 represents a vertical section of cupola or furnace used in my process, being a close cylindrical chamber with a flue, A, at top to carry off the carbonic-acid gas formed in decarbonizing the iron.
B is a small opening to receive the charge of fluid iron.
C C C are the tuyeres placed around the sides of the furnace, pointing downward at an angle such that they sweep about three-quarters of the bottom of the chamber, the muzzles of the tuyeres being about six inches above the bottom of the chamber.
D is a tap-hole for letting out the metal when refined. The chamber should not exceed three or four times the space occupied by the fluid iron. The blast is first let on into this chamber or cupola; then the fluid iron is poured in, which, by the cause hereinbefore described, commences a violent ebullition or boiling, which continues until the iron is sufficiently refined, when the tap-hole is opened and the metal let out.
What I claim as my invention or discovery, and desire to secure by Letters Patent, is—
Blowing blasts of air, either hot or cold, up and through a mass of liquid iron, the oxygen in the air combining with the carbon in the iron, causing a greatly increased heat and boiling commotion in the fluid mass and decarbonizing and refining the iron.
JAS. N. GRACY.
- 21 Aug - short biography, births, deaths and events on date of Kelly's birth.
- Henry Bessemer patent loss in U.S. to William Kelly - English reaction reported in Scientific American.
- William Kelly: a True History of the So-Called Bessemer Process, by John Newton Boucher. - book suggestion.




